Introduction:
Improved understanding of disease biology and treatment has transformed acute myeloid leukemia (AML) prognostication, as reflected in the most recent revision to the 2017 ELN risk classification in 2022. The key modifications include categorizing all FLT3-ITD mutated patients in the intermediate risk group, irrespective of allele frequency (AF), and classifying myelodysplasia-related mutations (MDSmut; ASXL1, BCOR, EZH2, RUNX1, SF3B1, SRSF2, STAG2, U2AF1, and ZRSR2) as adverse risk. Several cooperative groups have conducted studies to validate this new classification system. In this study, we compare the performance of the ELN 2017 and 2022 classifications in predicting the outcomes of a predominantly minority AML population.
Methods:
We conducted a retrospective review of patients >18 years old with newly diagnosed AML treated between July 2014 and February 2022 at Montefiore Medical Center, Bronx, NY. Patients' records were reviewed for demographics, mutations, cytogenetic abnormalities, therapy, and outcomes. Patients were risk-stratified according to both the ELN 2017 and 2022 criteria. Subjects with FLT3-ITD mutation with unknown allele frequency or who did not receive standard of care midostaurin were excluded. Rates of complete remission were assessed in both groups with ordinal regression and compared using Mann-Whitney U tests. Overall survival (OS) and event-free survival (EFS) were compared using Kaplan-Meier logistic regression.
Results:
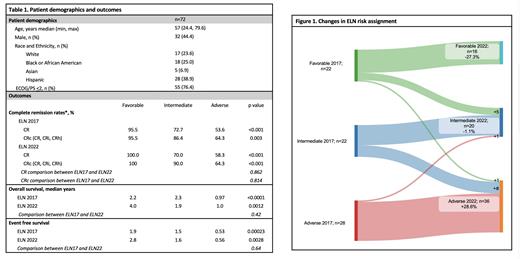
We identified 72 newly diagnosed AML patients treated with intensive induction chemotherapy. Thirty-two (44.4%) were male, with a median age at diagnosis of 57 years old (24.4-79.6). Seventeen (23.6%) were White, 18 (25.0%) were Black, and 5 (6.9%) were Asian. Twenty-eight (38.9%) identified as Hispanic. Fifty-five (76.4%) patients had ECOG/PS < 2. The median WBC on presentation was 9.75 (95% CL 5.7-20.8) cells/dL; median blast percentage on bone marrow biopsy was 52% (95% CL 35%-65%).
Updating risk classification from ELN17 to ELN22 decreased the favorable risk group from 22 (30.6%) to 16 (22.2%) with 5 patients re-classified due to FLT3-ITD mutations and 1 due to TP53 heterozygosity. The intermediate risk group decreased from 22 to 20 patients, with 4 FLT3-ITD patients reclassified from the favorable group, 1 patient with high FLT3-ITD AF from adverse group and 8 patients upclassified to adverse group due to MDSmut (STAG2 n=4, SF3B1 n=2, SRSF2 n=1, U2AF1 n=1) and cytogenetics (t(8;16) n=1). The adverse group increased from 28 (38.9%) to 36 (50%), predominantly due to myelodysplasia-related mutations.
CR rates in patients assigned by ELN17 were 95.5%, 72.7%, and 53.6% compared to 100.0%, 70.0%, 58.3% in the ELN22 (fav, int, adv) risk groups, respectively. Both scores were significant predictors of CR rates as measured via ordinal regression, p=<0.001 for both ELN17 and ELN22, respectively. There were no significant differences in the CR rates between ELN17 and ELN22 risk groups (p=0.862).
Reclassification according to ELN22 did not result in statistically significant differences in OS and EFS (p=0.42 and 0.64 respectively). Both risk models had statistically significant differences between risk categories. Risk stratification by ELN22 trended towards improved prediction of both OS and EFS with each increase in risk category, compared to ELN17. The median OS for the ELN17 groups was 2.2, 2.3, and 0.97 years respectively, while it was 4.0, 1.9, and 1.2 years for ELN22 (fav, int, adv) respectively. Similarly, EFS for patients classified by ELN22 followed a more predicted pattern than ELN17, with 1.9, 1.5, and 0.53 years for the three ELN17 groups and 2.8, 1.6 and 0.56 years for ELN22 groups (fav, int, adv) respectively. Combined outcome concordance values (c-index) were in the expected range for AML prediction scores at 0.76 and 0.77.
Conclusion:
Our study compares the performance of the ELN 2017 and 2022 risk classifications in a predominantly minority AML patient population. Limited by small numbers, we showed that, compared to ELN17, the ELN22 classification provided more predictable trends for CR, OS and EFS, but we were unable to detect statistically significant differences between the 2 scores' prediction. Continuous study of larger, inclusive patient cohorts will be important to validate the prognostication schemas in ethnically and racially diverse populations.
Disclosures
Shastri:Kymera Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Gilead Sciences: Membership on an entity's Board of Directors or advisory committees; Rigel Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees. Gritsman:iOnctura: Research Funding; ADC Therapeutics: Research Funding. Verma:Celgene: Consultancy; Throws Exception: Current equity holder in private company; Acceleron: Consultancy; Novartis: Consultancy; Eli Lilly: Research Funding; Medpacto: Research Funding; Incyte: Research Funding; GSK: Research Funding; Curis: Research Funding; Janssen: Honoraria; Bakx: Consultancy, Current equity holder in private company; Stelexis: Consultancy, Current equity holder in private company, Honoraria; Prelude: Research Funding; Bristol Myers Squibb: Research Funding. Konopleva:AbbVie, Forty Seven, Precision Biosciences, Gilead Sciences, Genentech, Janssen, Sanofi, MEI Pharma, Daiichi Sankyo Pharmaceutical, AstraZeneca Co., Menarini.: Consultancy; Reata Pharmaceuticals.: Current holder of stock options in a privately-held company, Patents & Royalties; Abbvie, Allogene Therapeutics, Cellectis, Forty Seven, Gilead Sciences, Genentech, Sanofi, MEI Pharma, Rafael Pharmaceuticals, Daiichi Sankyo Pharmaceutical, AstraZeneca Co., Menarini, Precision BioSciences.: Research Funding. Mantzaris:Kite, a Gilead company: Honoraria.